Governance
Data and Privacy
MedX is both HIPAA and GDPR compliant, and is an active manager of all requisite auditing as part of its governance processes. All HIPAA and GDPR compliance ensures that any and all personal and identifiable health data is kept private, secure, and used appropriately.

Certifications
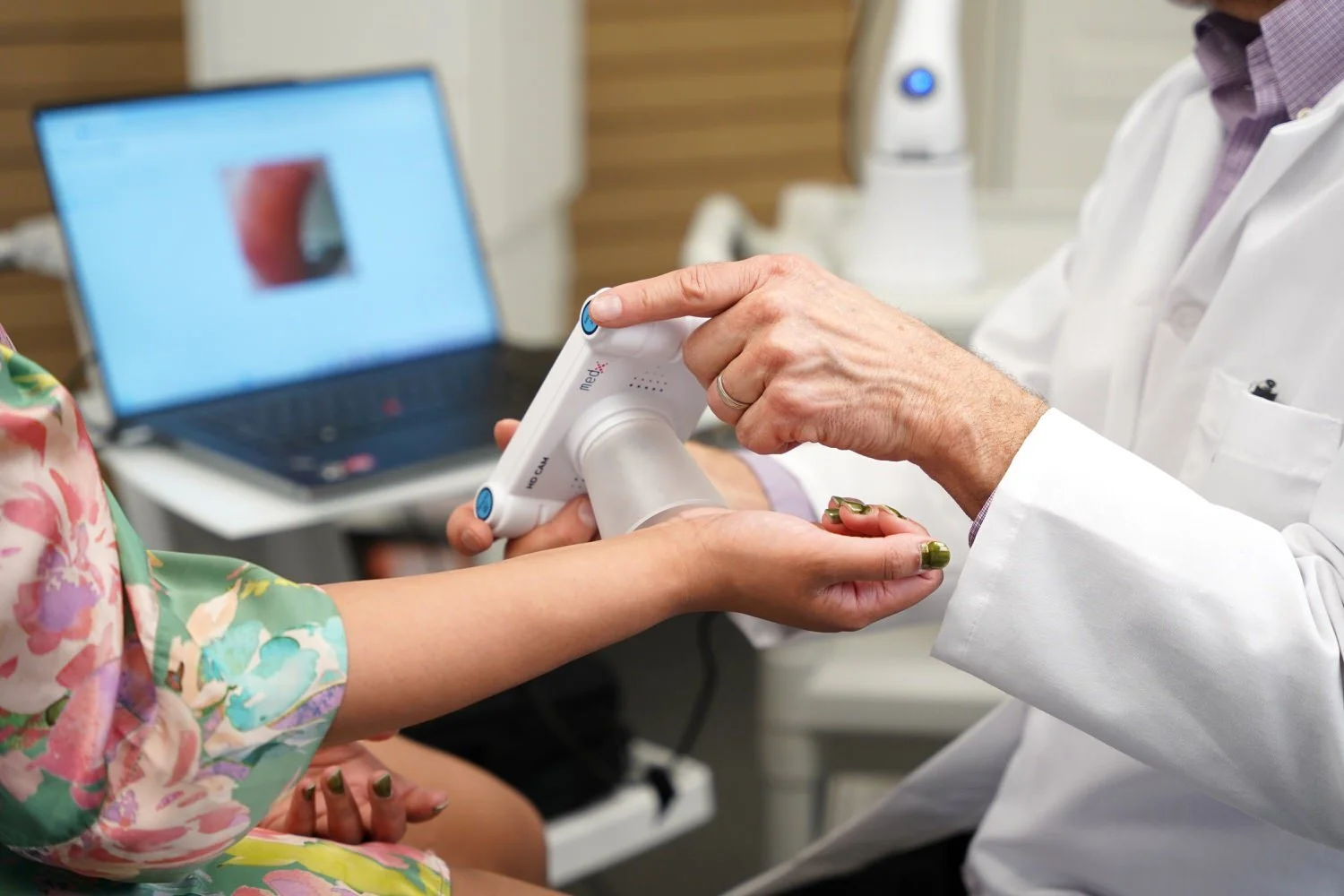
Operating with FDA and CE approvals, MedX hardware and Software are all classified as medical-devices and are licensed for operations across 38 territories, including Canada, USA, UK, Europe and Australia.

Interoperability
MedX’s DermSecure Platform is built on the FHIR HL7 backbone. FHIR (Fast Healthcare Interoperability Resources) is a standard developed by HL7 (Health Level Seven International).Our platform can connect with hospitals, labs, EHRs, and other systems that support FHIR. This allows for more seamless highly secure data sharing, where required.

Audit
MedX actively manages various audits across territory, device and operating system, including ISO13045 and MDSAP.




